


Transforming
rare disease ventures
into market winners



Trusted by Biotech and Pharma companies across Europe


A Partner Who Understands Your Reality
Rare disease environments require fast decisions, strong alignment and deep market knowledge. I’ve spent over 30 years working in exactly these conditions - supporting launches, building teams and navigating complex market access situations. Today, I work closely with Biotech and Pharma companies to bring that experience where it creates the most value.

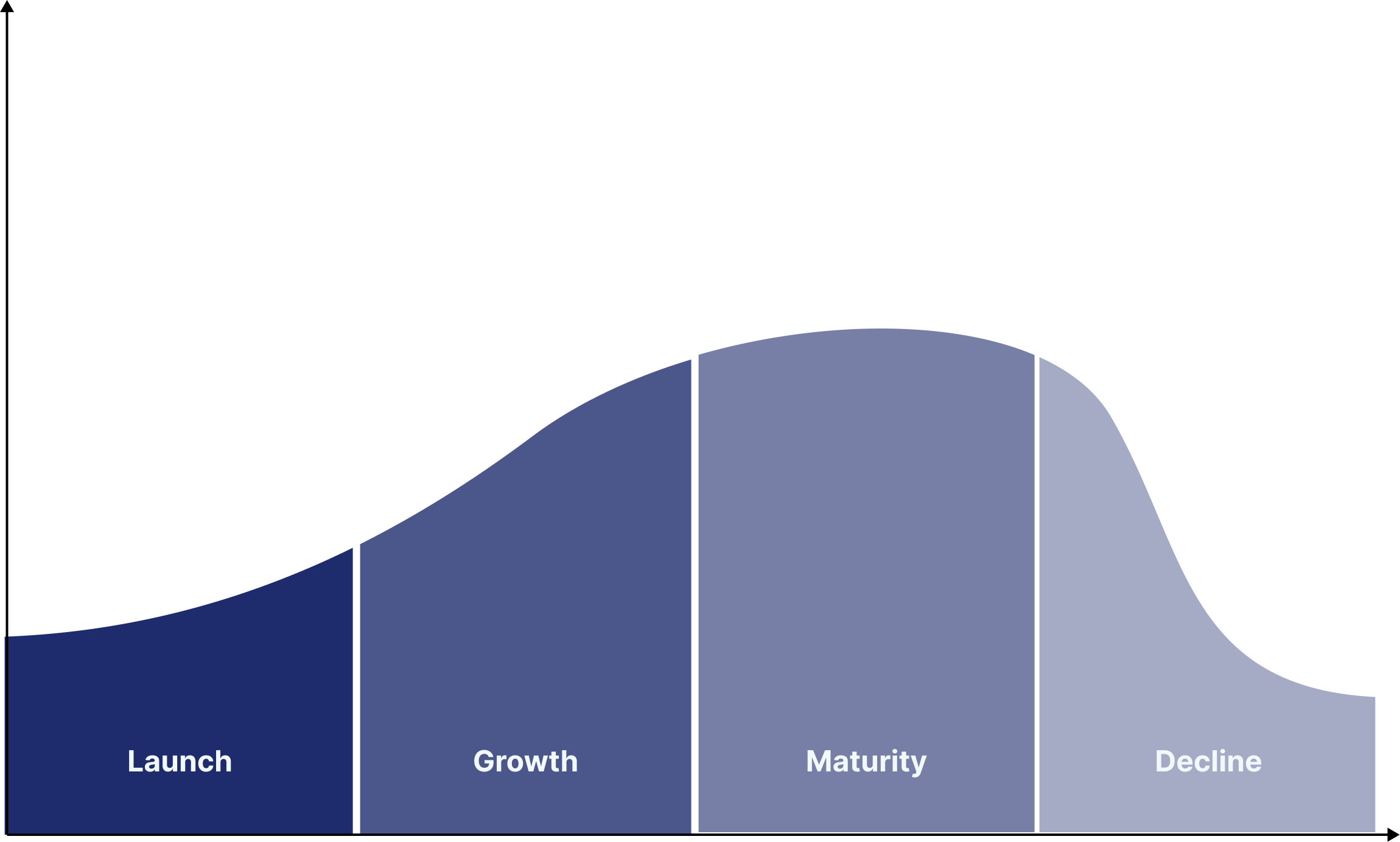
Addressing the Most Critical Challenges in Rare Disease Markets
I support rare disease companies across the full lifecycle - from early strategy and access planning to launch, growth and long-term value creation.
Market Access
Supporting Access Strategy from Early Planning to Successful Negotiation
Challenges
From Launch Preparation to Market Adoption in Highly Specialized Environments
Challenges
Go-to-Market
Team Development
Building Strong Teams and Leadership Structures That Deliver Results
Challenges
Creating Value Through Strategic Medical Initiatives and Trusted Partnerships
Challenges
Public-Private Partnerships

A Structured Way of Working in Complex Environments
WHat I bring to the table
My background combines senior leadership, rare disease expertise and real-world execution experience across multiple markets and organizations.

Building High-Performance Teams
With more than 20 years of leadership experience, I support organizations in developing strong teams, clear structures and a culture that delivers.
Proven Launch Experience in Rare Diseases
I have led 10 successful product launches - including 6 orphan drugs - across highly specialized and complex markets.
30+ Years Industry Expertise. 9 Years General Management.
Deep experience across Pharma and Biotech, with senior leadership roles in Germany, Austria and Switzerland.

Strong Industry Network & Negotiation Expertise
I bring long-standing relationships with key opinion leaders, industry partners and service providers - and the confidence to lead complex negotiations.

Beyond Quick Wins - Towards Sustainable Results
I support rare disease organizations with strategies that remain sustainable after launch decisions are made.
The Right Level of Support in High-Stakes Phases
Rare disease launches demand focus and speed. I offer flexible involvement tailored to your timeline, team and priorities.
Two Flexible Ways to Partner With Me
Depending on your situation, I can either step into your organization as an interim leader - or support you as an external project partner. Let's talk to find out what is the best solution for you.
Interim management role
I step into your organization as an operational leader during critical phases. This works when you have a leadership gap, a launch window that cannot wait, or a temporary vacancy in market access or commercial leadership. I become part of your team and drive daily execution.
Project partner and advisor
I work with you on a defined project or strategic question. This is best when your team is strong but needs rare-disease expertise, when you want an outside perspective on key decisions, or when you need fast delivery without adding headcount.

What they said
Real words from people I’ve worked with.
"Hannes combines rare-disease market expertise with something much harder to find: the ability to turn strategy into execution. What distinguishes Hannes is not only his experience, but also his judgement, reliability and pragmatic leadership style."

"Together we developed the concept of a clinical and research collaboration agreement between the company he has worked for and the University hospital aiming at becoming a leading Center of Excellence for BBS in Europe and beyond, which has now become a reality."

"He combines strong strategic thinking with a practical, hands-on approach to execution. Hannes navigates ambiguity well, takes ownership of challenges and consistently focuses on moving initiatives forward. He is also highly collaborative and communicates clearly, which made our cross-country coordination smooth and effective."

TTR Amyloidosis, Alnylam Pharmaceuticals, Zurich
"Hannes is a very experienced negotiator with clear strategies, excellent negotiation skills and with a strong network in and beyond Germany."

RECENT SUCCESSES
EMPOWERING RARE DISEASE COMPANIES
THROUGH INNOVATIVE MARKET CONSULTING
